Water Heater Repair Denver reduces contamination in water through a fine physical barrier or a chemical process. This helps clean water in food preparation, industrial manufacturing, and heating or cooling devices.
Boiling is one way of filtering but doesn’t remove chemicals or limescale. Other methods include ion exchange and distillation. Ion exchange filters swap magnesium and calcium ions with sodium ions in the water. This softens the water and prevents rust in equipment.
Water purification is important to ensure drinking water is safe, sustainable, and free from unpleasant taste and odor. It also helps to prevent disease-causing bacteria, which can be present in water sources. These bacteria can cause diarrhea, cholera, and other dangerous diseases that can affect millions of people around the world every year. Moreover, contaminated drinking water can also hurt the environment.
Water filters can purify drinking water for many reasons, including health concerns, environmental issues, and the need to meet certain nutrient requirements. To use water filters effectively, it is important to understand the types of contaminants that need to be removed from drinking water. This can help you decide which filter is best for your needs.
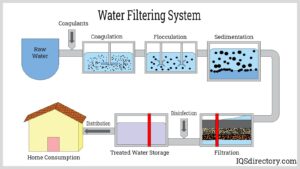
There are several water filtration methods, but the most common is using a physical barrier to remove contaminants from drinking water. Depending on the type of filter used, this may involve either a physical or chemical process. This method is widely used for various applications, including agricultural irrigation, public and private water supplies, and recreational uses.
A sand or ceramic filter is one of the most popular water filter methods. This is a simple and inexpensive way to remove impurities from drinking water. However, this method does not remove all harmful chemicals. It can leave behind small amounts of essential chemicals for human health, such as fluorine, necessary for healthy teeth.
Another common method of water filtration is membrane processes. These processes can be used to remove both primary and secondary pollutants. Secondary pollutants are less toxic than primary ones but still threaten the environment. They are derived from three main sources:
- Energy combustion.
- Reagents in some cleaning processes.
- Products that result from removing primary pollutants.
Another popular method of removing contaminants from drinking water is to boil it. This process eliminates most harmful drinking water substances and kills bacteria and other microorganisms that cause infection and illness. In addition, boiling water can be used to clean utensils and equipment.
Pretreatmenteatmentpretreatmentant step in the filtration process. It removes physical and chemical contaminants that interfere with filtration or damage equipment. These contaminants include sediment, bacteria, and chemicals. The pretreatment involves several steps, including screening to remove large particles, sand filtration, ion exchange, and membrane filtration. It also includes aeration and disinfection.
The primary goal of water pre-treatment is to reduce the concentrations of organic and inorganic matter that may affect human health and the environment. These contaminants can be found in groundwater, surface water, and wastewater. Industrial processes or natural events can introduce them to the water supply. Water pre-treatment pretreatment levels of these pollutants to make it safe for drinking and other uses.
There are various types of water filtration systems used in industrial applications. Carbon filters are commonly used because they capture all water chemicals and eliminate unwanted tastes and odors. They can filter to 1 micron and remove cysts like giardia and cryptosporidium. Other forms of filtration can also be used, such as the reverse osmosis filtration system, which effectively removes dissolved minerals.
This filtration method involves passing the water through a semi-permeable membrane at high pressure. This can remove ions and molecules, making it ideal for treating hard water. This method is also useful in reducing the level of calcium and magnesium in the water, which are known to cause scale build-up on equipment. Another method of removing these substances is the ion exchange technique, which involves exchanging magnesium and calcium for sodium ions. The ion exchange is accomplished through small bead resins that must be charged periodically.
Water pre-treatment is important for industrial uses. It helps to protect expensive machinery from foreign materials, which can clog pumps and membranes. This type of filtration also improves water quality for steam and vapor production. It reduces the risk of limescale build-up, which can lead to system failure and higher repair costs. Additionally, it can help to reduce the amount of energy needed to heat water to a temperature that is suitable for cooking.
Filtration is an important part of the water treatment process in the water industry. It helps reduce the amount of harmful particles in the water supply, which can cause disease and other health problems. In addition, it helps to control the flow of contaminants in the water supply. The importance of water filtration can not be overstated. Water filtration systems can be found in industrial, commercial, and residential settings. They are also used in the food, textile, and oil production industries. The main purpose of these systems is to remove sediments, dissolved solids, heavy metals, and other impurities from the water supply. These systems use different types of filtration media, including sand, ceramic, and cellulose fibers.
In a typical filtration plant, the water is first screened through sediment filters to reduce the amount of contaminants in the feedwater. A granular or membrane filtration system follows this. The latter is usually combined with pretreatment to ensure the efficiency of the filtration process.
These filtration systems produce high-purity water for pharmaceuticals, electronics, and other uses. They can reduce the levels of organic matter, nitrogen compounds, and phosphorus in the wastewater. However, they are not effective at removing bacterial matter. This is due to the lack of contact time between the wastewater and the filter medium. As a result, the effectiveness of a filtration system depends on the size of the filter media and the length of time it is in contact with the wastewater.
Besides removing bacteria and other organic matter, water filtration plants can also remove turbidity, acidity, and color from the water. In addition, they can help remove odors and unpleasant tastes from the water. This water can then be used for cooking, drinking, and other purposes. However, the water must be tested before it is consumed.
The final step in the filtration process is disinfection. This kills bacteriophages, protozoa, and viruses, bypassing the desalination process. The bactericidal treatment uses UV radiation, ozone, or chlorination.
To ensure the water meets regulatory standards, it must be tested by an independent laboratory. These tests can include several elements, such as total dissolved solids, alkalinity, pH, hardness, and chloride concentration. The results of these tests will determine whether the water is safe to drink.
Chemical compatibility is the ability of two or more chemicals to mix and interact without adversely affecting each other. Combined with incompatible chemicals, they may cause a violent reaction that can lead to hazardous fumes, explosions, or mechanical weakening. Proper dilution procedures should be followed when mixing chemicals to minimize the risk of incompatibility.
Chemical safety is an important aspect of water filtration. Using the correct equipment and following proper dilution procedures can help to ensure that dangerous reactions are avoided. However, it is also important to understand how different substances react with each other so that you can be prepared for any unforeseen hazards.
Considering chemical compatibility is especially important when working with hazardous chemicals. Incompatible chemicals can cause dangerous reactions that can harm workers and damage equipment. Incompatibilities can also cause environmental damage.
To reduce the risk of such reactions, it is important to read labels carefully and consult chemical compatibility charts and databases when selecting chemicals. The charts can provide information on the chemical reactivity of thousands of common hazardous chemicals and absorbents. These resources can help you determine which chemicals are compatible and should not be mixed.
When choosing chemicals for laboratory use, it is essential to consult the safety data sheet and compatibility chart before handling or storing them. The SDS contains detailed information on the properties and hazards of each chemical, including how it can react with other chemicals. The compatibility chart will indicate how safe a material can handle and store with other materials under specified conditions.
Chemical compatibility can be especially important in pharmaceutical manufacturing, where various chemicals are required. In one case, an incompatible combination of chemicals resulted in a chemical reaction that formed toxic byproducts and compromised the quality of the finished medication. This incident underscores the importance of careful consideration and testing regarding chemical compatibility in pharmaceutical manufacturing.
In addition to checking chemical compatibility, testing equipment with the chemicals used in the laboratory is important. This will allow you to see firsthand how the equipment performs and how it might react with specific chemicals. For example, knowing whether it will bind with the acid or release toxic gases is important if a membrane filter is used with an acidic solution.